2025-12-05
Crystalvue awarded the Gold Medal Award by Taiwan Bioindustry Association for the outstanding operational performance as a leading enterprise.
2024-11-01
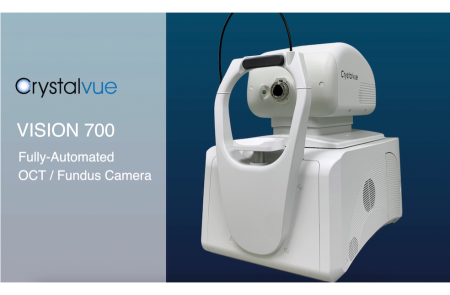
Crystalvue Vision 700, a fully automated OCT system integrating fundus photography, was awarded the Bronze in the Innovation in Technology Award at the 2024 Taipei Biotech Awards.
2024-07-20
Check Out Our New YouTube Video on the VISION 700! It's a fully-automated optical coherence tomography device integrated with a fundus camera.
2024-05-06
Crystalvue held a groundbreaking ceremony today for its new factory in Bade District, Taoyuan City. It is expected to be completed in the first half of 2026.
2024-04-08
Crystalvue will attend 2024 ESCRS in Spain from September 6th to 10th.
2023-05-16
The 41st Congress of the European Society of Cataract and Refractive Surgeons (ESCRS) will take place between the 8th-12th September 2023 in Vienna.
2023-05-16
Crystalvue will attend 2023 APACRS Annual Meeting at Singapore from June 8th to 10th.
2023-02-24
Crystalvue is thrilled to announce that it has been awarded the prestigious Taiwan BIO Awards. Crystalvue's CEO, Dr. William Wang, had the privilege of meeting with President Tsai Ing-wen.
2022-07-12
Crystalvue set a new monthly revenue record in June, with a monthly increase of 39% and an annual increase of 64% due to strong and steady sales in Europe and North America.
2022-02-14
President Tsai meets the winners of 2021 Taiwan Bio Award. As the Gold Medal winner, Crystalvue General Manager, Mr. CP Chuang, was invited to meet President Tsai.
2021-11-22
CV-700 is an Automated Patternless Workstation of Lens that combines tracing, edging, beveling, grooving, and blocking functions.
2021-11-22
With steady increase in new orders and new product launch, Crystalvue is optimistic about the business outlook and targets double digit growth in 2022.